
We specialize in producing advanced pharmaceutical APIs tailored to meet diverse healthcare needs. Our portfolio includes innovative products like Nefopam Hydrochloride, Sugammadex Sodium, and Midazolam, all developed through cutting-edge research and stringent quality controls. By prioritizing safety, efficacy, and global regulatory compliance, we deliver APIs that redefine industry standards and support a healthier tomorrow.
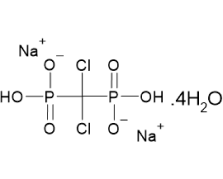
Clodronic acid (INN), also known as Clodronate disodium (USAN), is a first-generation, non-nitrogenous bisphosphonate used for the prevention and treatment of osteoporosis. It is particularly effective in postmenopausal women and men for reducing vertebral fractures and managing conditions like hyperparathyroidism, hypercalcemia in malignancy, multiple myeloma, and fracture-related pain. Clodronic acid also exhibits potent anti-inflammatory and analgesic effects, with a marked reduction in inflammatory markers such as IL-1β, IL-6, and TNF-α.
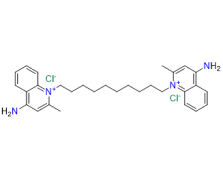
Dequalinium Chloride is a cationic bola amphiphilic compound composed of two quinaldinium rings linked by 10 methylene groups. This versatile quaternary ammonium compound is widely used for its antimicrobial properties, effectively treating mouth, throat infections, and vaginal candidiasis. Beyond its antimicrobial use, it plays a crucial role in reducing inflammation and enhancing drug delivery systems.
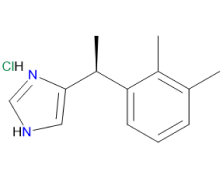
Dexmedetomidine Hydrochloride is a highly selective alpha-2 adrenoreceptor agonist known for its sedative, analgesic, and anxiolytic properties with minimal respiratory depression. As an imidazole derivative, it exerts its effects by binding to alpha-2 adrenoceptors in the brain, inhibiting norepinephrine release from synaptic vesicles, thereby reducing sympathetic activity. This mechanism results in effective sedation, analgesia, and anxiety reduction, making it a vital drug in clinical settings, particularly in intensive care and procedural sedation.
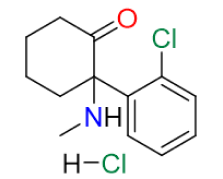
Ketamine Hydrochloride is a versatile pharmaceutical agent, classified as a cyclohexanone, where the 2-position hydrogen is substituted with a 2-chlorophenyl group and a methylamino group. Known for its role as a NMDA receptor antagonist, Ketamine Hydrochloride is widely used for its potent anesthetic and analgesic effects. It is a critical component in medical and veterinary anesthesia, as well as pain management, owing to its unique properties and fast-acting nature.
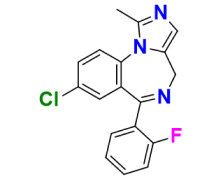
Midazolam is a short-acting hypnotic-sedative with anxiolytic and amnestic properties, widely used in procedures like dentistry, cardiac surgery, and anesthesia. Its short action and cardiorespiratory stability make it ideal for elderly and high-risk patients. Administered through various routes (oral, intravenous, intramuscular, nasal, or buccal), its primary effects include sedation, anxiety reduction, muscle relaxation, amnesia, and anticonvulsant activity, making it essential in medical settings.
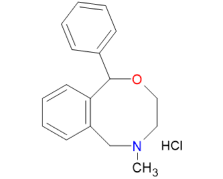
Nefopam Hydrochloride is a powerful non-opioid analgesic, known for its efficacy in relieving acute and chronic pain, including post-operative, dental, musculoskeletal, traumatic, and cancer pain. Our approach to the synthesis and production of Nefopam Hydrochloride sets us apart, ensuring superior quality, safety, and efficiency at every step.
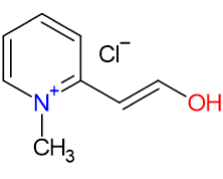
Pralidoxime Chloride is a cholinesterase reactivator used as an antidote for organophosphate poisoning, commonly caused by pesticides and nerve agents. This prescription medicine is vital in treating poisoning cases where muscle weakness or respiratory depression occurs. It works by reactivating the enzyme acetylcholinesterase, which has been inactivated by these toxic agents. This reactivation breaks down the excess acetylcholine caused by the poisoning, helping to reverse muscle weakness and restore respiratory function.
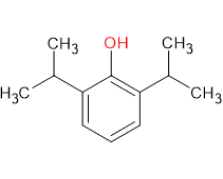
Propofol is an essential anesthetic widely used in surgeries, from minor to complex procedures, ensuring patient comfort and safety. It is commonly administered during endoscopies, colonoscopies, and various diagnostic imaging tests that require patient sedation to minimize discomfort and anxiety. Additionally, Propofol plays a critical role in managing life-threatening conditions like refractory status epilepticus.
Remifentanil Hydrochloride is a potent, short-acting synthetic opioid analgesic used primarily during surgery to provide effective pain relief and as an adjunct to anesthesia. Its rapid onset and short duration of action make it ideal for surgical settings, ensuring precise control of pain management.
Rocuronium Bromide is a short-acting, non-depolarizing steroidal neuromuscular blocker commonly used alongside general anesthesia to facilitate endotracheal intubation and provide muscle relaxation during surgery or mechanical ventilation. Given its effect on neuromuscular function, a reversal agent, like Sugammadex, is often required to address potential residual paralysis post-procedure.
Sevoflurane is a widely used volatile anesthetic for inducing and maintaining general anesthesia. Known for its sweet smell, non-flammable nature, and rapid onset of action, Sevoflurane is particularly valued for not irritating the airways, making it ideal for patient comfort. As a highly fluorinated methyl isopropyl ether, it has become a preferred choice in medical anesthesia.
Sugammadex Sodium is a groundbreaking drug in the class of selective relaxant binding agents (SRBA), providing a safer, more efficient reversal of neuromuscular blockade caused by agents like Rocuronium and Vecuronium in general anesthesia. With significant peri-operative benefits, Sugammadex offers a superior solution for managing muscle relaxation during surgery.

Desflurane is a highly fluorinated methyl ethyl ether used for the maintenance of general anesthesia. It stands out among volatile anesthetic agents due to its rapid onset and offset, allowing precise control over anesthesia depth. With its low blood solubility, Desflurane ensures quick induction and recovery, making it particularly beneficial for outpatient surgeries and procedures requiring fast post-operative recovery. This property makes it a preferred choice in modern anesthetic practices, providing effective anesthesia at lower concentrations while enabling swift patient turnaround.

Pridinol Mesylate is a muscle relaxant used to alleviate muscle spasms and stiffness. As an anticholinergic agent, it works by blocking the neurotransmitter acetylcholine, which plays a key role in muscle contractions. By inhibiting acetylcholine activity, Pridinol Mesylate effectively reduces muscle spasms, promotes relaxation, and provides pain relief, making it an essential therapeutic option in musculoskeletal disorders.

Clodronic acid (INN), also known as Clodronate disodium (USAN), is a first-generation, non-nitrogenous bisphosphonate used for the prevention and treatment of osteoporosis. It is particularly effective in postmenopausal women and men for reducing vertebral fractures and managing conditions like hyperparathyroidism, hypercalcemia in malignancy, multiple myeloma, and fracture-related pain. Clodronic acid also exhibits potent anti-inflammatory and analgesic effects, with a marked reduction in inflammatory markers such as IL-1β, IL-6, and TNF-α.

Dequalinium Chloride is a cationic bola amphiphilic compound composed of two quinaldinium rings linked by 10 methylene groups. This versatile quaternary ammonium compound is widely used for its antimicrobial properties, effectively treating mouth, throat infections, and vaginal candidiasis. Beyond its antimicrobial use, it plays a crucial role in reducing inflammation and enhancing drug delivery systems.

Dexmedetomidine Hydrochloride is a highly selective alpha-2 adrenoreceptor agonist known for its sedative, analgesic, and anxiolytic properties with minimal respiratory depression. As an imidazole derivative, it exerts its effects by binding to alpha-2 adrenoceptors in the brain, inhibiting norepinephrine release from synaptic vesicles, thereby reducing sympathetic activity. This mechanism results in effective sedation, analgesia, and anxiety reduction, making it a vital drug in clinical settings, particularly in intensive care and procedural sedation.

Ketamine Hydrochloride is a versatile pharmaceutical agent, classified as a cyclohexanone, where the 2-position hydrogen is substituted with a 2-chlorophenyl group and a methylamino group. Known for its role as a NMDA receptor antagonist, Ketamine Hydrochloride is widely used for its potent anesthetic and analgesic effects. It is a critical component in medical and veterinary anesthesia, as well as pain management, owing to its unique properties and fast-acting nature.

Midazolam is a short-acting hypnotic-sedative with anxiolytic and amnestic properties, widely used in procedures like dentistry, cardiac surgery, and anesthesia. Its short action and cardiorespiratory stability make it ideal for elderly and high-risk patients. Administered through various routes (oral, intravenous, intramuscular, nasal, or buccal), its primary effects include sedation, anxiety reduction, muscle relaxation, amnesia, and anticonvulsant activity, making it essential in medical settings.

Nefopam Hydrochloride is a powerful non-opioid analgesic, known for its efficacy in relieving acute and chronic pain, including post-operative, dental, musculoskeletal, traumatic, and cancer pain. Our approach to the synthesis and production of Nefopam Hydrochloride sets us apart, ensuring superior quality, safety, and efficiency at every step.

Pralidoxime Chloride is a cholinesterase reactivator used as an antidote for organophosphate poisoning, commonly caused by pesticides and nerve agents. This prescription medicine is vital in treating poisoning cases where muscle weakness or respiratory depression occurs. It works by reactivating the enzyme acetylcholinesterase, which has been inactivated by these toxic agents. This reactivation breaks down the excess acetylcholine caused by the poisoning, helping to reverse muscle weakness and restore respiratory function.

Propofol is an essential anesthetic widely used in surgeries, from minor to complex procedures, ensuring patient comfort and safety. It is commonly administered during endoscopies, colonoscopies, and various diagnostic imaging tests that require patient sedation to minimize discomfort and anxiety. Additionally, Propofol plays a critical role in managing life-threatening conditions like refractory status epilepticus.

Remifentanil Hydrochloride is a potent, short-acting synthetic opioid analgesic used primarily during surgery to provide effective pain relief and as an adjunct to anesthesia. Its rapid onset and short duration of action make it ideal for surgical settings, ensuring precise control of pain management.

Rocuronium Bromide is a short-acting, non-depolarizing steroidal neuromuscular blocker commonly used alongside general anesthesia to facilitate endotracheal intubation and provide muscle relaxation during surgery or mechanical ventilation. Given its effect on neuromuscular function, a reversal agent, like Sugammadex, is often required to address potential residual paralysis post-procedure.

Sevoflurane is a widely used volatile anesthetic for inducing and maintaining general anesthesia. Known for its sweet smell, non-flammable nature, and rapid onset of action, Sevoflurane is particularly valued for not irritating the airways, making it ideal for patient comfort. As a highly fluorinated methyl isopropyl ether, it has become a preferred choice in medical anesthesia.

Sugammadex Sodium is a groundbreaking drug in the class of selective relaxant binding agents (SRBA), providing a safer, more efficient reversal of neuromuscular blockade caused by agents like Rocuronium and Vecuronium in general anesthesia. With significant peri-operative benefits, Sugammadex offers a superior solution for managing muscle relaxation during surgery.

Lorem ipsum dolor sit amet consectetur. Egestas hac sit risus diam vitae sed tincidunt. At sit diam cursus fusce.

Troikaa Pharmachem Pvt. Ltd.
Registered Office:
Troikaa House,
Satya Marg, Bodakdev,
Ahmedabad-380 054. Gujarat, India.
Corporate Identification Number: U24299GJ2021PTC127098
Copyright © 2025 Troikaa. All rights reserved.
Our Clodronate Disodium Tetrahydrate is synthesized through a validated, efficient process that ensures robust control of impurities, minimal cycle time, and a well-defined design space. By implementing QbD principles, we guarantee a high-quality API with consistent performance. We adhere to stringent ICH guidelines for impurity control, ensuring that our product meets the highest pharmaceutical standards for safety, reliability, and efficacy.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
The synthesis of Clodronate Disodium Tetrahydrate is built on HAZOP-driven safety practices. By systematically assessing risks and implementing preventive measures, we ensure a secure and efficient process. Coupled with advanced safety features and environmental safeguards, our operations prioritize safety at every stage.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | |
| 02 | Degradation impurities | 1 | Phosphonic acid derivatives (Phosphite ion) |
| 03 | Genotoxic impurities | 0 | |
| 04 | Process related impurities | 5 | Tetraisopropyl methylene diphosphonate, Tetraisopropyl dichloromethylene diphosphonate, Sodium chloride, Monoisopropyl, MonoChloro impurity |
| 05 | Aggregated impurities | 0 | |
| 06 | Literature impurities | 0 | |
| 07 | Nitrosamine impurities | 0 | |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 |
Choosing our Dequalinium Chloride means selecting a product developed with the utmost care, precision, and commitment to quality. Our advanced QbD-driven process, combined with rigorous impurity control and compliance with international standards, ensures a product that is safe, reliable, and consistent. Whether for treating infections or as part of more complex drug delivery systems, our Dequalinium Chloride is the ideal solution for pharmaceutical companies looking for the best in class.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
For Dequalinium Chloride, HAZOP provides a structured methodology to preemptively manage safety risks in production. By embedding this approach into our operations, we ensure a hazard-free process. Fail-safes for critical parameters, combined with thorough risk assessments and regular audits, uphold our commitment to safety and environmental stewardship.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 0 | -- |
| 03 | KSM impurities | 1 | 4-hydroxy-2-methylquinoline |
| 04 | Genotoxic impurities | 5 | Impurity-D, Impurity-E, Impurity-F and 4-chloro-2-methylquinoline, Benzene |
| 05 | Process related impurities | 3 | Impurity-A, Impurity-B and Impurity-C |
| 06 | Aggregated impurities | 0 | -- |
| 07 | Literature impurities | 3 | Determination of Dequalinium chloride and related impurities in cosmetics and pharmaceuticals by reversed-phase HPLC, A nuclear magnetic resonance method for the determination of the purity of commercial dequalinium chloride |
| 08 | Nitrosamine impurities | 1 | N-Nitrosodiethylamine |
| 09 | Elemental impurities | 24 | As per ICH Q3D (R2) |
| 10 | Isomer impurities | 0 | -- |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
Our Dexmedetomidine Hydrochloride API offers superior quality and safety through a QbD-driven four-step synthesis process, ensuring consistent Pharmacopoeial grade production. With stringent impurity control and full compliance with ICH guidelines for Nitrosamine, Genotoxic, and Elemental impurities, it provides a reliable and trusted solution for pharmaceutical manufacturers, making it ideal for clinical applications in sedation and analgesia.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
In manufacturing Dexmedetomidine Hydrochloride, HAZOP plays a vital role in preventing hazards and enhancing safety. Through systematic risk identification, advanced safety measures, and regular parameter audits, we ensure a secure operational environment. HAZOP enables us to consistently deliver high-quality, safe, and compliant pharmaceutical solutions.
| Sr. No. | Types of Impurities | Number of Impurities | Common Names of Impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 1 | Dexmedetomidine oxide |
| 03 | KSM impurities | 0 | -- |
| 04 | Genotoxic impurities | 1 | Benzene |
| 05 | Process related impurities | 0 | -- |
| 06 | Aggregated impurities | 0 | -- |
| 07 | Literature impurities (USP monograph) | 5 | Hydroxymedetomidine, N-Benzyl Hydroxymedetomidine, Ethylmedetomidine, N-Benzyl medetomidine, N-Benzyl vinyl analogue |
| 08 | Nitrosamine impurities | 0 | -- |
| 09 | Elemental impurities | 24 | As per ICH Q3D (R2) |
| 10 | Isomer impurities | 1 | Levomedetomidine hydrochloride |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
By combining advanced synthesis techniques, rigorous impurity control, and a commitment to regulatory compliance, we offer a Ketamine Hydrochloride API that stands out for its reliability, safety, and quality. Our five-step synthesis process, optimized through the QbD approach, ensures consistent delivery of a Pharmacopoeial grade product that meets the needs of a wide range of medical applications.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
The production of Ketamine Hydrochloride is guided by HAZOP principles, ensuring all safety concerns are anticipated and mitigated. This proactive framework allows us to maintain a fail-safe production process, complemented by advanced safety audits and robust risk management strategies, delivering a secure and compliant product.
| Sr. No. | Types of Impurities | Number of Impurities | Common Names of Impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 0 | -- |
| 03 | KSM impurities | 2 | o-Chlorobenzaldehyde, o-Chlorobenzotrichloride |
| 04 | Genotoxic impurities | 4 | Chlorocyclopentane, 2-CPK, DBE, Benzene |
| 05 | Process related impurities | 3 | Impurity-A, Impurity-B, and Impurity-C |
| 06 | Aggregated impurities | 0 | -- |
| 07 | Literature impurities | 3 | Synthesis of ketamine from a nontoxic procedure: a new and efficient route. The novel methoxetamine analogs N-ethylnorketamine hydrochloride (NENK), 2-MeO-N-ethylketamine hydrochloride (2-MeO-NEK), and 4-MeO-N-ethylketamine hydrochloride (4-MeO-NEK) elicit rapid antidepressant effects via activation of AMPA and 5-HT2 receptors. |
| 08 | Nitrosamine impurities | 1 | N-Nitrosoketamine |
| 09 | Elemental impurities | 24 | As per ICH Q3D (R2) |
| 10 | Isomer impurities | 0 | -- |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
By combining advanced synthetic techniques, stringent impurity control, and a commitment to regulatory excellence, our Midazolam API stands out for its unparalleled quality, safety, and reliability. Our process is designed to meet the highest Pharmacopoeial standards, making it a trusted choice for pharmaceutical manufacturers worldwide.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Midazolam production integrates HAZOP as a cornerstone of our safety strategy. By identifying and mitigating risks at every stage, we maintain the highest safety standards. Comprehensive safety audits, critical parameter monitoring, and environmental safeguards ensure Midazolam manufacturing remains secure, efficient, and fully compliant with industry regulations.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 2 | N-Oxide, Hydroxy-methyl midazolam |
| 03 | Genotoxic impurities | 2 | Dihydroquinazoline 3-oxide, quinazoline 3-Oxide |
| 04 | Process related impurities | 3 | Desfluoromidazolam, Dihydromidazolam, Aminomethylbenzodiazepine |
| 05 | Aggregated impurities | 1 | Midazolam Dimer |
| 06 | Literature impurities | 11 | OPRD 2023, US7776852B2, Acta Pharm. 2013 |
| 07 | Nitrosamine impurities | 4 | N-Nitroso dihydromidazolam, N-Nitroso reduced midazolam, N-Nitroso N-Oxide Nitromethylene, N-Nitroso aminomethylbenzodiazepine |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 1 | Midazolam 6-H isomer |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
Our commitment to advanced process innovation, rigorous safety protocols, and strict regulatory compliance ensures that Nefopam Hydrochloride produced by us not only meets but exceeds industry standards. By leveraging sophisticated technologies and scientific excellence, we provide a product that is both safer and more efficient, offering unparalleled value to our customers.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Safety is paramount in the pharmaceutical industry, and we go beyond compliance to ensure our processes are the safest in the market. Through comprehensive risk assessments and the integration of advanced safety features, our Nefopam Hydrochloride synthesis process is built to prevent hazards at every stage. We employ HAZOP, a systematic approach to identifying and mitigating potential risks in our processes. This proactive measure allows us to anticipate and address safety concerns before they arise, ensuring a robust and fail-safe operational environment. Additionally, fail-safes for critical parameters and regular safety audits are conducted, ensuring both personnel safety and environmental protection.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 1 | N-Oxide |
| 03 | Genotoxic impurities | 3 | IPMS, EO and Benzene |
| 04 | Process related impurities | 4 | Nefomide, Nefodiol, Deoxy, Ketomaine |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 4 | OPRD 2017 |
| 07 | Nitrosamine impurities | 3 | NDMA, N-Nitroso MMEA, N-Nitroso Desmethyl Nefopam |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurities (Deletion & Addition impurity) | 0 | -- |
Our Pralidoxime Chloride API is manufactured in a state-of-the-art facility with multi-kilogram production capacity. All raw materials are sourced from GMP-compliant suppliers, ensuring product integrity and quality. The result is a highly efficient, consistent, and safe API that meets global pharmaceutical standards.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
HAZOP forms the foundation of our safety measures for Pralidoxime Chloride production. This systematic approach enables us to identify and address potential risks proactively, ensuring a safe manufacturing process. Regular safety audits and critical parameter monitoring further reinforce our commitment to operational excellence and environmental protection.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | - |
| 02 | Degradation impurities | 0 | - |
| 03 | Genotoxic impurities | 1 | Dimethyl sulphate |
| 04 | Process related impurities | 05 | Picolinic acid, Pyridine-2-aldehyde, Pyridine-2-aldoxime, Hydroxylamine sulphate, Isopropyl chloride |
| 05 | Aggregated impurities | 0 | - |
| 06 | Literature impurities | 5 | J Pharm Sci. 1986 Jun;75(6):608-11. doi: 10.1002/jps.2600750618. |
| 07 | Nitrosamine impurities | 0 | - |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 1 | Pralidoxime anti-isomer |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | - |
Our Propofol API is distinguished by its optimized synthesis process, stringent impurity control, and high reproducibility. We maintain strict adherence to regulatory standards, ensuring that our Propofol is safe, reliable, and ideal for use in a variety of medical applications.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Propofol manufacturing is anchored in HAZOP principles, ensuring a hazard-free and secure production process. By integrating advanced risk assessment methodologies and fail-safe mechanisms, we maintain the highest safety standards. Routine audits and a commitment to compliance guarantee safe and reliable operations.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | KSM impurities | 3 | Phenol, Salicylic acid, 4-Hydroxy phthalic acid |
| 02 | Degradation impurities | 0 | - |
| 03 | Genotoxic impurities | 0 | -- |
| 04 | Process related impurities | 6 |
4-hydroxy-3-isopropyl benzoic acid 4-isopropoxy-3,5-diisopropyl benzoic acid 4-isopropoxy-3-isopropyl benzoic acid 4,4’-oxy dibenzoic acid Isopropyl-4-isopropoxy benzoate Methyl-4-hydroxy-3,5-diisopropyl benzoate |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Specified impurities | 6 |
Ph. Eur. Imp G Ph. Eur. Imp E Ph. Eur. Imp J Ph. Eur. Imp K Ph. Eur. Imp L Ph. Eur. Imp O |
| 07 | Unspecified impurities | 9 |
Ph. Eur. Imp A Ph. Eur. Imp B Ph. Eur. Imp C Ph. Eur. Imp D Ph. Eur. Imp F Ph. Eur. Imp H Ph. Eur. Imp I Ph. Eur. Imp N Ph. Eur. Imp P |
| 08 | Nitrosamine impurities | 0 | -- |
| 09 | Elemental impurities | 24 | As per ICH Q3D |
| 10 | Isomer impurities | 0 | -- |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
Our Remifentanil Hydrochloride API stands out for its superior quality and consistent production. The use of the QbD approach ensures a robust and reproducible manufacturing process, with stringent control over impurities to guarantee compliance with ICH guidelines. This makes our Remifentanil Hydrochloride a trusted and reliable option for pharmaceutical manufacturers, delivering high performance in surgical pain management and anesthesia applications.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Remifentanil Hydrochloride production prioritizes safety through HAZOP, a structured approach to risk management. This process identifies potential hazards early and incorporates fail-safes to mitigate them. With regular audits and comprehensive safety protocols, we uphold an environment of security and compliance throughout production.
| Sr. No. | Types of Impurities | Number of Impurities | Common Names of Impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 1 | Impurity C |
| 03 | KSM impurities | 0 | -- |
| 04 | Genotoxic impurities | 1 | Benzene |
| 05 | Process related impurities | 0 | -- |
| 06 | Aggregated impurities | 0 | -- |
| 07 | Literature impurities | 15 | Impurity-A, Impurity-B, Impurity-C, Impurity-D, Impurity-E, Impurity-F, Impurity-G, Impurity-H, Impurity-I, Impurity-J, Impurity-K, Impurity-L, Impurity-M, Impurity-N, Impurity-O |
| 08 | Nitrosamine impurities | 0 | -- |
| 09 | Elemental impurities | 24 | As per ICH Q3D (R2) |
| 10 | Isomer impurities | 0 | -- |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
With our cutting-edge synthesis process, stringent impurity control, and a robust commitment to regulatory compliance, we deliver a Rocuronium Bromide API that offers superior performance and safety. Our focus on quality ensures that every batch meets the highest industry standards, making it the preferred choice for pharmaceutical manufacturers seeking reliability and excellence.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Our Rocuronium Bromide API is designed to exceed regulatory expectations. Through a combination of advanced process controls and state-of-the-art manufacturing technologies, we achieve a Pharmacopoeial grade product that stands out for its purity and consistency. Our commitment to Quality by Design (QbD), continuous process monitoring, and compliance with international regulatory standards makes us a trusted partner in the pharmaceutical industry.
The synthesis of Rocuronium Bromide is fortified by HAZOP, enabling us to proactively detect and address potential risks. This rigorous process ensures operational safety, supported by advanced safety features and routine audits. With HAZOP at the core, we deliver a reliable and safe product while safeguarding both personnel and the environment.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | - |
| 02 | Degradation impurities | 0 | - |
| 03 | Genotoxic impurities | 1 | Allyl bromide |
| 04 | Process related impurities | 4 | O-allyl Rocuronium, 3-Acetyl Rocuronium Bromide, Desacetyl Rocuronium Bromide, Bis-Allyl Rocuronium |
| 05 | Aggregated impurities | 0 | - |
| 06 | Literature impurities | 8 | J. Chromatogr. A 2007 |
| 07 | Nitrosamine impurities | 0 | - |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | - |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | - |
Our Sevoflurane API stands out for its high-quality, safety, and compliance with stringent Pharmacopoeial standards. With an optimized synthesis process driven by QbD and DoE, we ensure consistent product quality and comprehensive impurity control. Our commitment to process safety further enhances reliability, making our Sevoflurane an ideal choice for medical use.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Sevoflurane production leverages HAZOP to identify and mitigate risks proactively. This structured safety approach ensures a robust, fail-safe process, complemented by regular safety audits and advanced environmental safeguards. HAZOP enables us to deliver a secure and compliant product with uncompromising quality.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | KSM impurities | 2 | Hexafluoro acetone, Hexafluoro propylene oxide (HFPO) |
| 02 | Degradation impurities | 0 | - |
| 03 | Genotoxic impurities | 0 | -- |
| 04 | Process related impurities | 1 | Sevoflurane Acetal |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Specified impurities | 3 |
Sevoflurane Related Compound A Sevoflurane Related Compound B Sevoflurane Related Compound C |
| 07 | Unspecified impurities | 1 | Sevochlorane |
| 08 | Nitrosamine impurities | 0 | -- |
| 09 | Elemental impurities | 24 | As per ICH Q3D |
| 10 | Isomer impurities | 0 | -- |
| 11 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
Our advanced approach to Sugammadex Sodium manufacturing integrates deep scientific expertise, meticulous process development, and adherence to stringent regulatory guidelines. With a focus on ensuring the highest quality, safety, and regulatory compliance, our Sugammadex Sodium offers a trusted, high-performance solution for the medical community.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
In producing Sugammadex Sodium, we employ HAZOP to meticulously assess and address potential safety risks. This structured approach ensures robust safety protocols across the process, preventing hazards before they occur. Coupled with regular audits and advanced fail-safe mechanisms, we deliver a safe and sustainable production environment that aligns with global safety standards.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 3 | Dia-1 of Sulfoxide, Dia-2 of Sulfoxide, Disulfide |
| 03 | Genotoxic impurities | 1 | Benzene |
| 04 | Process related impurities | 2 | Mono bromo & Mono hydroxyl impurity |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 2 | Chromatography, 2022 J Pharm Biomed Anal, 2022 |
| 07 | Nitrosamine impurities | 1 | NDMA |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurities (Deletion & Addition impurity) | 0 | -- |
Our Desflurane API is developed with an advanced synthesis process emphasizing process safety, impurity control, and regulatory compliance. By implementing cutting-edge manufacturing techniques, we ensure a highly pure, safe, and reliable product, meeting the stringent demands of the pharmaceutical industry. Our expertise in optimizing synthesis and impurity control allows us to deliver a product that ensures both patient safety and operational efficiency.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Our Desflurane manufacturing process is refined using Quality by Design (QbD) principles, ensuring consistency and reproducibility at a commercial scale. The process has been carefully optimized to enhance efficiency and quality, with a well-defined design space that allows for precise control over manufacturing variables. Critical Process Parameters (CPPs), Critical Quality Attributes (CQAs), and Critical Material Attributes (CMAs) have been thoroughly evaluated to enhance process reliability and minimize variability. The result is a robust and well-controlled synthesis process that meets stringent regulatory and pharmacopoeial standards, ensuring a high-purity final product.
We ensures Desflurane’s process safety through a HAZOP, identifying and mitigating risks in synthesis. By controlling critical parameters like temperature, pressure, and reaction kinetics, we prevent hazards and ensure operational integrity. Fail-safe mechanisms, real-time monitoring, and routine safety audits enhance reliability, ensuring compliance with global standards while minimizing impurities and safeguarding both personnel and product quality.
Desflurane synthesis prioritizes impurity control, ensuring compliance with pharmaceutical quality standards. Thorough risk assessments minimize nitrosamine and mutagenic impurities, enhancing clinical safety. Genotoxic Impurities (GTIs) are rigorously evaluated, and process optimizations eliminate harmful byproducts. Adhering to ICH Q3D guidelines, elemental impurities are strictly controlled, ensuring high purity, efficacy, and patient safety.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 0 | -- |
| 03 | Genotoxic impurities | 1 | Benzene |
| 04 | Process related impurities | 7 | Isoflurane, MDC, Acetone, DCFM, TCFM, TCTFE, Chloroform |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 7 | OPRD 2011 |
| 07 | Nitrosamine impurities | 0 | -- |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
Our Pridinol Mesylate API is developed with a meticulously optimized synthesis process, ensuring high purity, reproducibility, and compliance with stringent regulatory standards. By leveraging advanced process control strategies, we deliver a product that meets the highest pharmaceutical quality benchmarks, ensuring both efficacy and patient safety.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
The synthesis of Pridinol Mesylate is developed using the Quality by Design (QbD) approach, ensuring a robust and well-controlled process. During development, Critical Process Parameters (CPPs), Critical Material Attributes (CMAs), and Critical Quality Attributes (CQAs) were carefully identified and addressed to guarantee consistency in product quality. Given the sensitivity of Pridinol Mesylate to moisture, pH, and temperature, the purification process was precisely optimized to maintain stability and achieve the desired product specifications.
Our HAZOP approach ensures safety, risk mitigation, and regulatory compliance in Pridinol Mesylate manufacturing. Potential hazards related to moisture, pH, temperature fluctuations, and impurity formation are thoroughly assessed and controlled. Advanced monitoring systems, fail-safe mechanisms, and routine safety audits enhance operational integrity. Comprehensive training ensures personnel preparedness, ensuring high-quality, safe, and compliant Pridinol Mesylate production.
Pridinol Mesylate synthesis ensures stringent impurity control, maintaining exceptional purity and safety. Rigorous purification minimizes Genotoxic, Nitrosamine, and Elemental Impurities, complying with global regulatory standards. Advanced process optimization prevents impurity formation, ensuring high stability. Adhering to ICH Q3D guidelines, our Pridinol Mesylate API guarantees superior quality, reliability, and patient safety.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 1 | N-Oxide |
| 03 | Genotoxic impurities | 1 | Benzene |
| 04 | Process related impurities | 2 | Impurity B, Ene impurity |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 3 | JPBA, 2008 |
| 07 | Nitrosamine impurities | 1 | NPIP |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |