Lorem ipsum dolor sit amet consectetur. In id quis dolor turpis enim dui dictum a lorem. Non faucibus amet ac ultrices. Purus risus quis urna bibendum venenatis lobortis.
Thank you for taking the time to visit our website.


Innovation drives our Research & Development efforts, creating solutions that shape the future of healthcare. Our dedicated R&D team specializes in drug discovery, process optimization, and novel formulation development. With cutting-edge technology and expertise, we transform ideas into impactful products, delivering excellence and advancing pharmaceutical science.



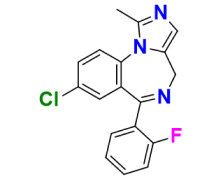
Midazolam is a short-acting hypnotic-sedative with anxiolytic and amnestic properties, widely used in procedures like dentistry, cardiac surgery, and anesthesia. Its short action and cardiorespiratory stability make it ideal for elderly and high-risk patients. Administered through various routes (oral, intravenous, intramuscular, nasal, or buccal), its primary effects include sedation, anxiety reduction, muscle relaxation, amnesia, and anticonvulsant activity, making it essential in medical settings.

Lorem ipsum dolor sit amet consectetur. Egestas hac sit risus diam vitae sed tincidunt. At sit diam cursus fusce.

Troikaa Pharmachem Pvt. Ltd.
Registered Office:
Troikaa House,
Satya Marg, Bodakdev,
Ahmedabad-380 054. Gujarat, India.
Corporate Identification Number: U24299GJ2021PTC127098
Copyright © 2025 Troikaa. All rights reserved.
By combining advanced synthetic techniques, stringent impurity control, and a commitment to regulatory excellence, our Midazolam API stands out for its unparalleled quality, safety, and reliability. Our process is designed to meet the highest Pharmacopoeial standards, making it a trusted choice for pharmaceutical manufacturers worldwide.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Midazolam production integrates HAZOP as a cornerstone of our safety strategy. By identifying and mitigating risks at every stage, we maintain the highest safety standards. Comprehensive safety audits, critical parameter monitoring, and environmental safeguards ensure Midazolam manufacturing remains secure, efficient, and fully compliant with industry regulations.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 2 | N-Oxide, Hydroxy-methyl midazolam |
| 03 | Genotoxic impurities | 2 | Dihydroquinazoline 3-oxide, quinazoline 3-Oxide |
| 04 | Process related impurities | 3 | Desfluoromidazolam, Dihydromidazolam, Aminomethylbenzodiazepine |
| 05 | Aggregated impurities | 1 | Midazolam Dimer |
| 06 | Literature impurities | 11 | OPRD 2023, US7776852B2, Acta Pharm. 2013 |
| 07 | Nitrosamine impurities | 4 | N-Nitroso dihydromidazolam, N-Nitroso reduced midazolam, N-Nitroso N-Oxide Nitromethylene, N-Nitroso aminomethylbenzodiazepine |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 1 | Midazolam 6-H isomer |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | -- |
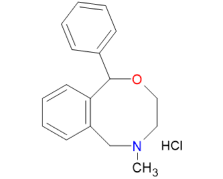
Our commitment to advanced process innovation, rigorous safety protocols, and strict regulatory compliance ensures that Nefopam Hydrochloride produced by us not only meets but exceeds industry standards. By leveraging sophisticated technologies and scientific excellence, we provide a product that is both safer and more efficient, offering unparalleled value to our customers.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Safety is paramount in the pharmaceutical industry, and we go beyond compliance to ensure our processes are the safest in the market. Through comprehensive risk assessments and the integration of advanced safety features, our Nefopam Hydrochloride synthesis process is built to prevent hazards at every stage. We employ HAZOP, a systematic approach to identifying and mitigating potential risks in our processes. This proactive measure allows us to anticipate and address safety concerns before they arise, ensuring a robust and fail-safe operational environment. Additionally, fail-safes for critical parameters and regular safety audits are conducted, ensuring both personnel safety and environmental protection.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 1 | N-Oxide |
| 03 | Genotoxic impurities | 3 | IPMS, EO and Benzene |
| 04 | Process related impurities | 4 | Nefomide, Nefodiol, Deoxy, Ketomaine |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 4 | OPRD 2017 |
| 07 | Nitrosamine impurities | 3 | NDMA, N-Nitroso MMEA, N-Nitroso Desmethyl Nefopam |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurities (Deletion & Addition impurity) | 0 | -- |
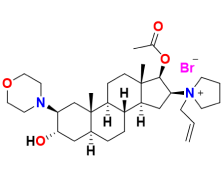
With our cutting-edge synthesis process, stringent impurity control, and a robust commitment to regulatory compliance, we deliver a Rocuronium Bromide API that offers superior performance and safety. Our focus on quality ensures that every batch meets the highest industry standards, making it the preferred choice for pharmaceutical manufacturers seeking reliability and excellence.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
Our Rocuronium Bromide API is designed to exceed regulatory expectations. Through a combination of advanced process controls and state-of-the-art manufacturing technologies, we achieve a Pharmacopoeial grade product that stands out for its purity and consistency. Our commitment to Quality by Design (QbD), continuous process monitoring, and compliance with international regulatory standards makes us a trusted partner in the pharmaceutical industry.
The synthesis of Rocuronium Bromide is fortified by HAZOP, enabling us to proactively detect and address potential risks. This rigorous process ensures operational safety, supported by advanced safety features and routine audits. With HAZOP at the core, we deliver a reliable and safe product while safeguarding both personnel and the environment.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | - |
| 02 | Degradation impurities | 0 | - |
| 03 | Genotoxic impurities | 1 | Allyl bromide |
| 04 | Process related impurities | 4 | O-allyl Rocuronium, 3-Acetyl Rocuronium Bromide, Desacetyl Rocuronium Bromide, Bis-Allyl Rocuronium |
| 05 | Aggregated impurities | 0 | - |
| 06 | Literature impurities | 8 | J. Chromatogr. A 2007 |
| 07 | Nitrosamine impurities | 0 | - |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | - |
| 10 | Truncated Impurity (Deletion & Addition impurity) | 0 | - |
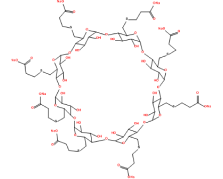
Our advanced approach to Sugammadex Sodium manufacturing integrates deep scientific expertise, meticulous process development, and adherence to stringent regulatory guidelines. With a focus on ensuring the highest quality, safety, and regulatory compliance, our Sugammadex Sodium offers a trusted, high-performance solution for the medical community.
Precision in API pharmaceutical manufacturing – where Innovation meets Excellence.M
In producing Sugammadex Sodium, we employ HAZOP to meticulously assess and address potential safety risks. This structured approach ensures robust safety protocols across the process, preventing hazards before they occur. Coupled with regular audits and advanced fail-safe mechanisms, we deliver a safe and sustainable production environment that aligns with global safety standards.
| Sr. No. | Types of impurities | Number of impurities | Common names of impurities |
|---|---|---|---|
| 01 | Unknown impurities | 0 | -- |
| 02 | Degradation impurities | 3 | Dia-1 of Sulfoxide, Dia-2 of Sulfoxide, Disulfide |
| 03 | Genotoxic impurities | 1 | Benzene |
| 04 | Process related impurities | 2 | Mono bromo & Mono hydroxyl impurity |
| 05 | Aggregated impurities | 0 | -- |
| 06 | Literature impurities | 2 | Chromatography, 2022 J Pharm Biomed Anal, 2022 |
| 07 | Nitrosamine impurities | 1 | NDMA |
| 08 | Elemental impurities | 24 | As per ICH Q3D |
| 09 | Isomer impurities | 0 | -- |
| 10 | Truncated Impurities (Deletion & Addition impurity) | 0 | -- |